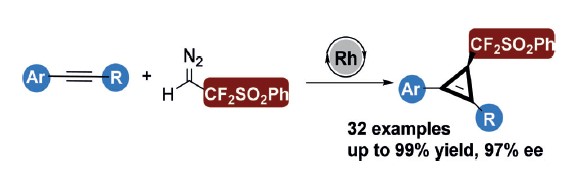
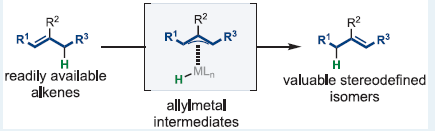
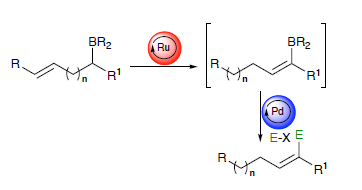
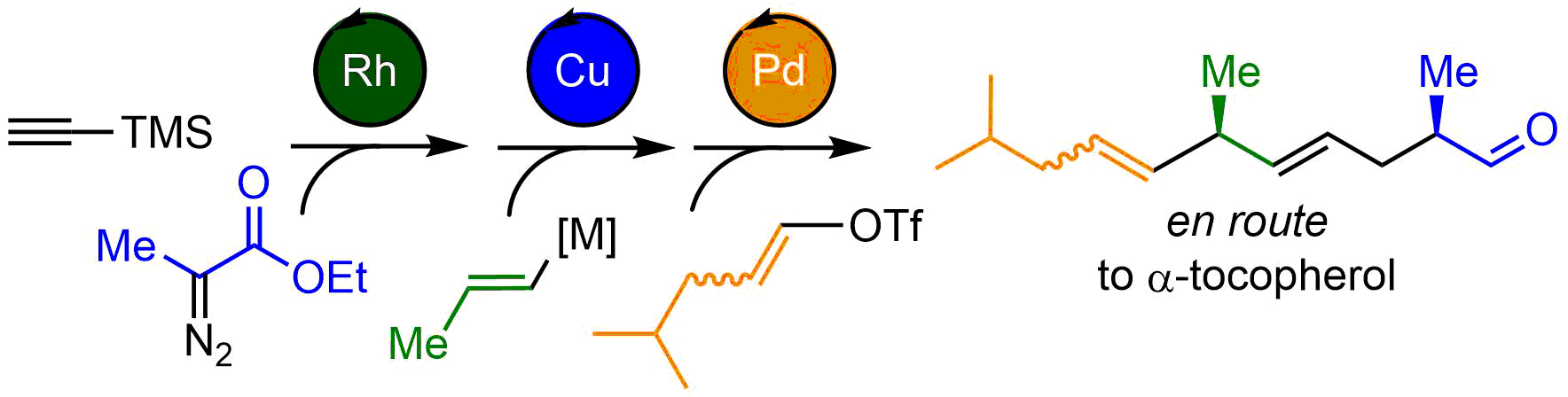
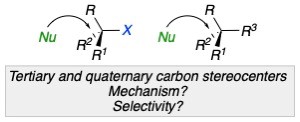
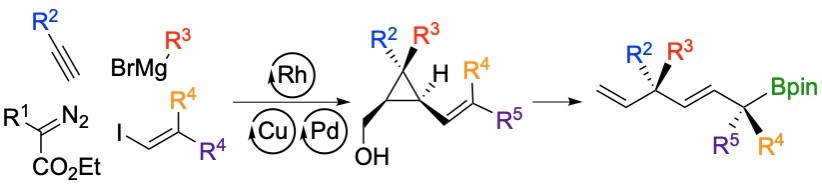
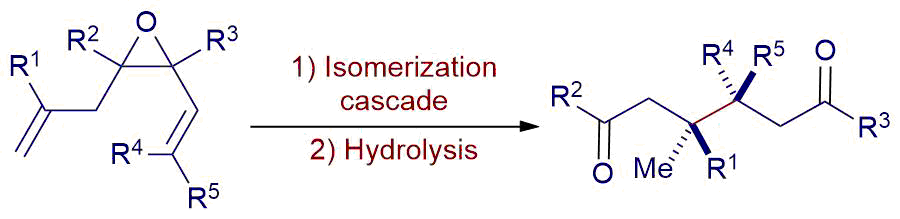
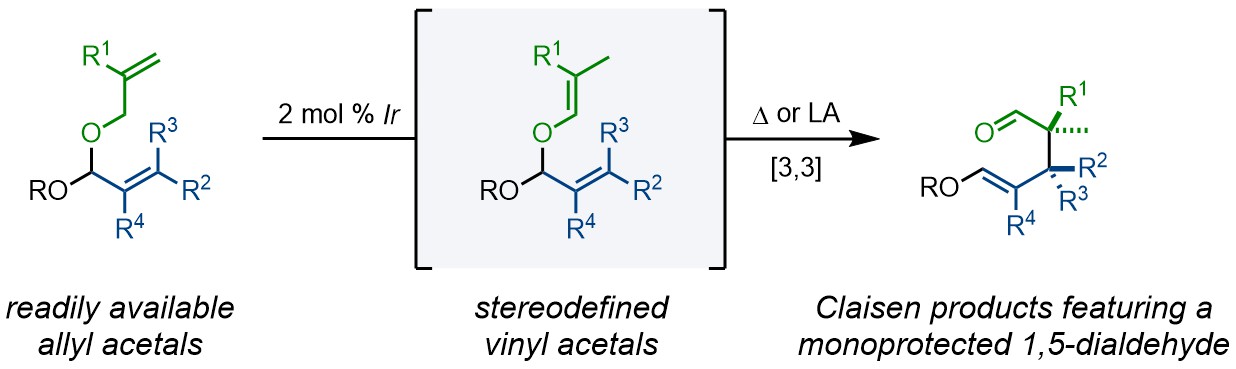
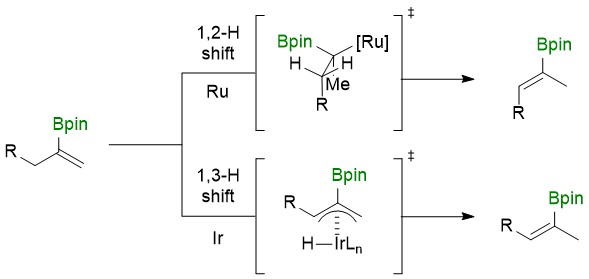
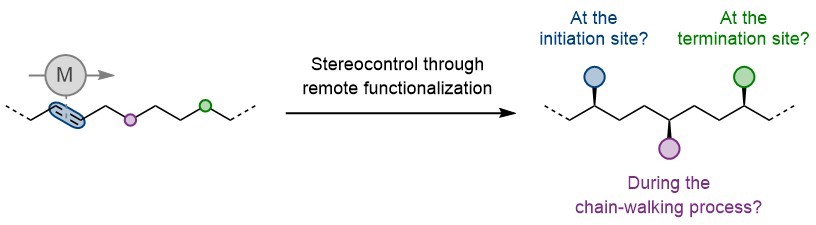
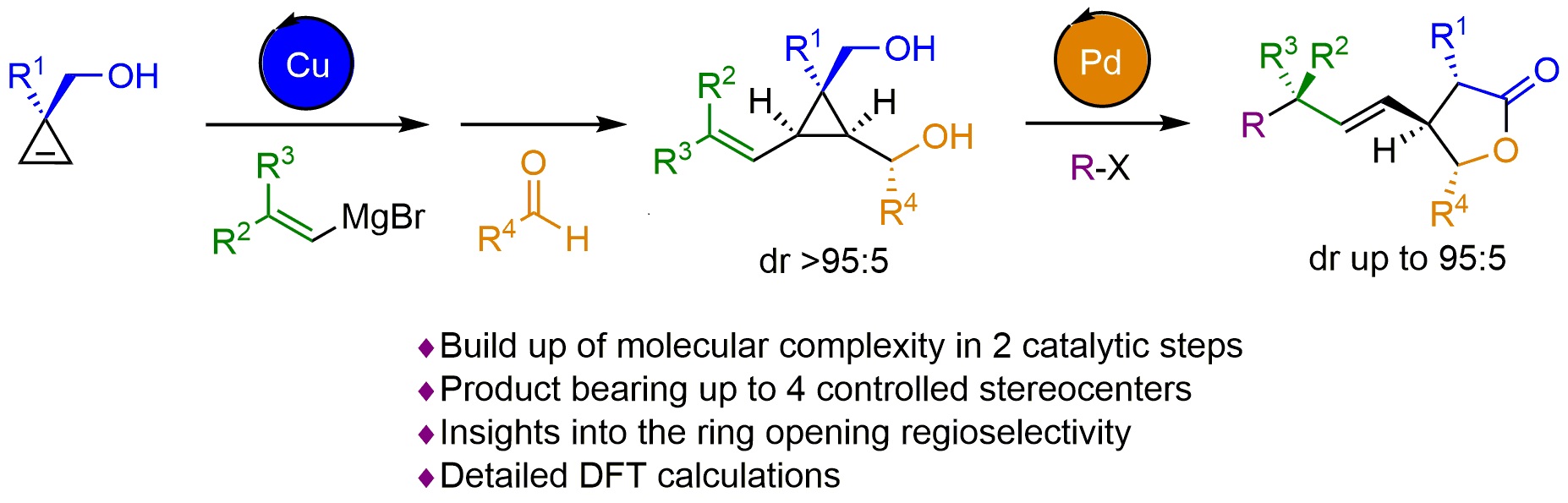
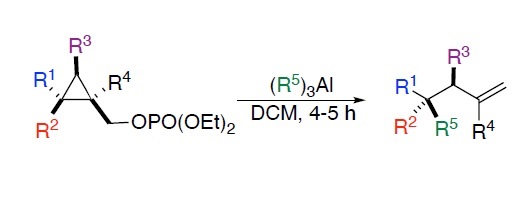
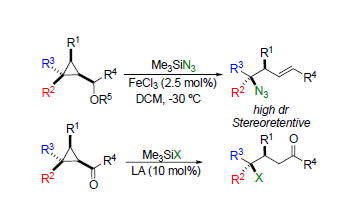
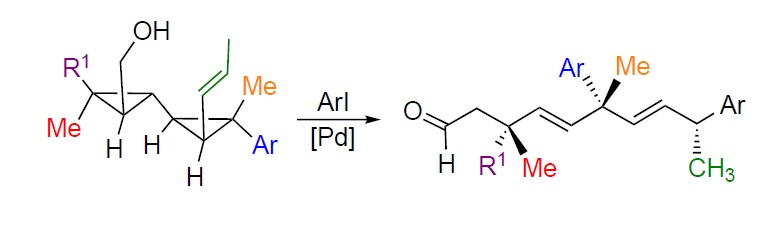
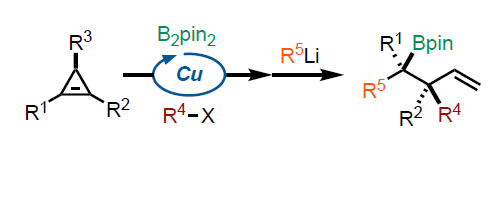
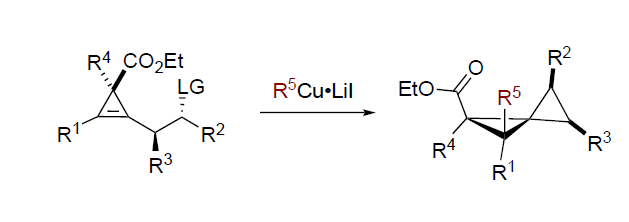
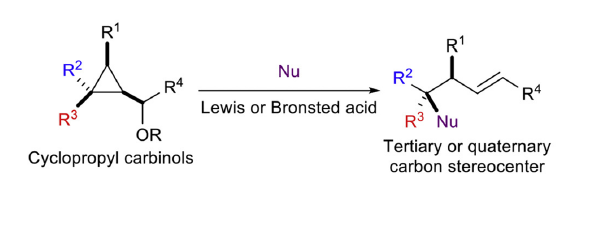
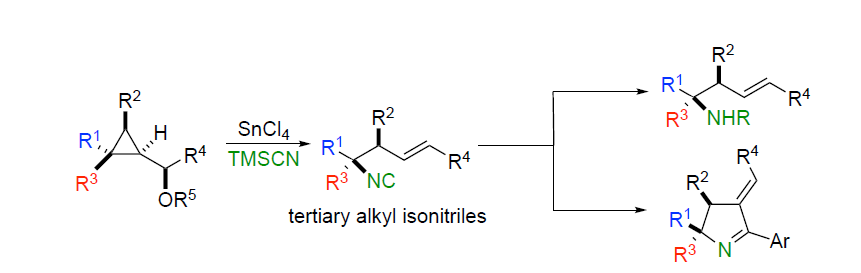
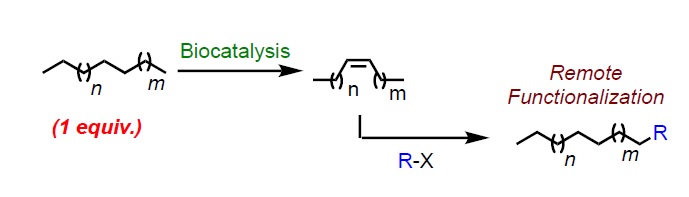
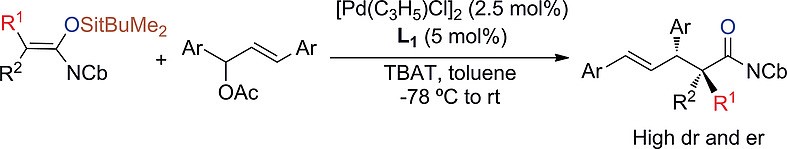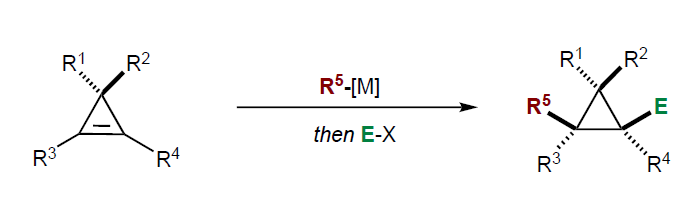Summary of the project
C–H functionalization has represented a paradigm shift from the standard logic of organic synthesis. Instead of focusing on orchestration of selective reactions at functional groups, C-H activation reactions have relied on the controlled functionalization of specific C–H bonds, even in the presence of more reactive functional groups. Since the pioneering work of Bergman, the field has matured to the point where it can now be considered a practical strategy for the synthesis of complex target molecules strongly impacting the field of fine chemical synthesis, materials science and pharmaceutical industry. However, the selective activation of non-functionalized alkanes has puzzled chemists for centuries and is always referred to one of the remaining major challenges in synthetic chemistry (Equation 1). Alkanes, or saturated hydrocarbons are the major constituents of natural gas and petroleum, but there are very few practical processes for converting alkanes directly to valuable products: “Alkanes are a primary source of fuel, but they also constitute a significant fraction of the carbon pool that is potentially accessible for chemical synthesis. It is ironic that such an abundant resource is presently so difficult to utilize“. A few decades after Bergman’s initial statement, this has yet to be achieved.
 Alkanes – also called paraffin meaning ‘not enough affinity – are inert molecules and converting this cheap and widely available hydrocarbon feedstock into added-value intermediates would tremendously affect the field of chemistry. Alkanes do react at high temperature, as encountered in combustion, but these reactions are usually difficult to control, and the only synthetic use of industrial significance for long-chain alkanes is the cracking process, which breaks large into smaller alkanes, a highly energy-consuming and non-selective process. Therefore, the activation of aliphatic alkanes possessing long alkyl chains has remained a vastly unsolved issue, as the number of potential sites of functionalization increases with the number of carbons. Only alkanes of small molecular weight (mainly C1 to C4) could be converted into more valuable commodities and synthetic intermediates (such as methanol, ethylene, propylene, butadiene). In contrast to the situation with methane, for longer saturated hydrocarbons, one must distinguish between non-equivalent but chemically very similar alkane substrate C−H bonds, and if the goal is functionalization at the terminus (as desired for most valuable applications), one must favour activation of the stronger, primary, C−H bonds at the expense of weaker and numerous secondary C-H bonds. Achieving a single chemical functionalization of a long hydrocarbon chain seems a priori an almost impossible task, as it requires overcoming several challenges including site selectivity. In recent years, although very few pioneering milestones have been achieved in the field of catalytic and homogeneous transition-metal based alkane activation, a general transformation of linear alkanes into a large variety of functionalized products remain completely impossible. How could we functionalize linear alkanes by using a single and unified new strategy? How could we selectively functionalize the terminus position favoring activation of the stronger primary C−H bonds? How could we design the synthesis of challenging molecular skeleton from simple linear saturated hydrocarbons and introduce many different functionalities (i.e. amine, nitrile, carboxylic acid, alkyne, alkene, allene, aldehyde, boron, silicon, sulfide, sulfoxide, sulfone, halide including fluorination, alcohol, alkyl, phosphonate)? How could alkane derivatives be the source of, at will, allyl or vinyl metal species?
Alkanes – also called paraffin meaning ‘not enough affinity – are inert molecules and converting this cheap and widely available hydrocarbon feedstock into added-value intermediates would tremendously affect the field of chemistry. Alkanes do react at high temperature, as encountered in combustion, but these reactions are usually difficult to control, and the only synthetic use of industrial significance for long-chain alkanes is the cracking process, which breaks large into smaller alkanes, a highly energy-consuming and non-selective process. Therefore, the activation of aliphatic alkanes possessing long alkyl chains has remained a vastly unsolved issue, as the number of potential sites of functionalization increases with the number of carbons. Only alkanes of small molecular weight (mainly C1 to C4) could be converted into more valuable commodities and synthetic intermediates (such as methanol, ethylene, propylene, butadiene). In contrast to the situation with methane, for longer saturated hydrocarbons, one must distinguish between non-equivalent but chemically very similar alkane substrate C−H bonds, and if the goal is functionalization at the terminus (as desired for most valuable applications), one must favour activation of the stronger, primary, C−H bonds at the expense of weaker and numerous secondary C-H bonds. Achieving a single chemical functionalization of a long hydrocarbon chain seems a priori an almost impossible task, as it requires overcoming several challenges including site selectivity. In recent years, although very few pioneering milestones have been achieved in the field of catalytic and homogeneous transition-metal based alkane activation, a general transformation of linear alkanes into a large variety of functionalized products remain completely impossible. How could we functionalize linear alkanes by using a single and unified new strategy? How could we selectively functionalize the terminus position favoring activation of the stronger primary C−H bonds? How could we design the synthesis of challenging molecular skeleton from simple linear saturated hydrocarbons and introduce many different functionalities (i.e. amine, nitrile, carboxylic acid, alkyne, alkene, allene, aldehyde, boron, silicon, sulfide, sulfoxide, sulfone, halide including fluorination, alcohol, alkyl, phosphonate)? How could alkane derivatives be the source of, at will, allyl or vinyl metal species?
This research is the basis of this new ERC research grant